The solution to a problem in chemical pre-treatment during anodising can also be simple sometimes. For example, the level of the bath can be significant for good or insufficient colouring, if we look at electrolytic colouring with tin salts. Ideally, the electrolytic tin dye bath (e.g. Alficolor 677) produces homogeneous bronze to black coloured aluminium surfaces.
But what happened if, even though the bath values were correct, you suddenly only get light-bronze and possibly even stained surfaces? Here it is worth taking a look at the bath level. Sometimes it happens that the bath level of the anodising bath is below the bath level of the colour bath and that part of the material is still sticking out of the anodising bath surface (for example the top profile layer). If this error is overlooked and the material subsequently enters the colour bath, faulty colouring will be the result.
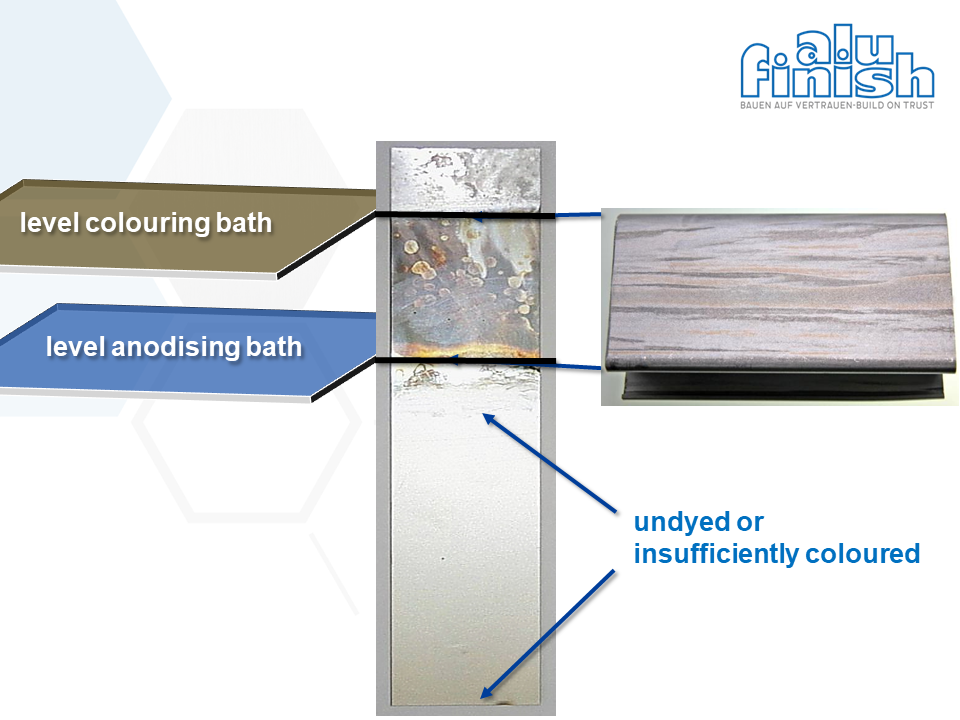
Why does this happen?
In our example, the top profile layer is not anodised and therefore enters the subsequent electrolytic colouring bath as a metallic surface. If the colouring process is carried out in the usual way, a large part of the applied electricity flows ineffectively over the metallic surface (without oxide layer). In these areas, there is then a more or less strong metallic tin deposition on the surface, while there is no longer enough electricity to colour the rest of the anodised product.
This results in a patchy and grey-brown upper profile layer, while profiles underneath are not coloured at all or only weakly coloured.

In order to simply avoid this error, it should therefore always be ensured that only completely anodised materials enter the colouring bath. This ensures that the colouring electricity can only be used for colouring and does not flow over metallic surface areas.
